Live streaming on Altcast.TV is now available!
FLUORIDE 🚱 POISON ON TAP 🎞️ FULL DOCUMENTARY
https://fluoridealert.org/news..../fluoride-poison-on-
STORY AT-A-GLANCE
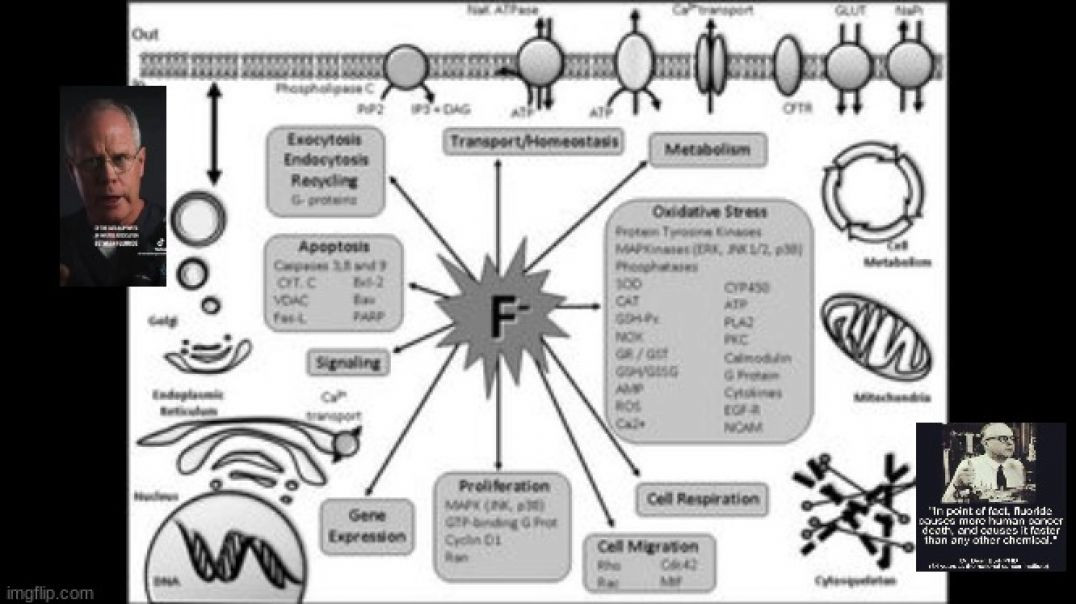
Since 1945, it has been widely accepted in the U.S. that fluoride is “safe and effective” to prevent tooth decay, but many health experts and scientists disagree
Rates of dental fluorosis among children and adolescents has spiked in recent years and, if fluoridated water is used, formula-fed infants are at particular risk for fluorosis even before their teeth are fully formed
The Fluoride Action Network has brought a lawsuit against the U.S. Environmental Protection Agency seeking to ban the deliberate addition of fluoride to American water supplies
By Dr. Mercola
[Read original article online]
https://articles.mercola.com/sites/articles/archive/2017/10/14/fluoride-poison-on-tap-documentary.aspx?utm_source=dnl&utm_medium=email&utm_content=art1&utm_campaign=20171014Z1_UCM&et_cid=DM161510&et_rid=84284116
Today, 74 percent of Americans on community water systems receive fluoridated water.1 Since 1945, it has been widely accepted in the U.S. that fluoride is “safe and effective” to prevent tooth decay. But is it really?
The 2015 documentary, “Fluoride: Poison on Tap,” seeks to expose what may be one of the longest-running and most successful deceptions known to mankind — adding industrial waste, in the form of fluoride, to public drinking water. You may be shocked at the lengths to which corporations, industry and government have gone to make this industrial waste product appear beneficial to your health.
Fluoride = Health: How Did We Get Here?
You may be surprised to know the first American commercial use of fluoride, in the form of sodium fluoride, was to kill insects, lice, mice and other vermin. It was quite effective. In the 1930s, aluminum-industry giant Alcoa was the largest producer of fluoride, releasing vapors into the atmosphere that crippled or killed farm animals and scorched crops and other vegetation. In those early years, many lawsuits were brought against Alcoa to recover damages from lost animals and crops.
Growing concerns about the seemingly negative effects of fluoride gas on human beings motivated the company to devise a means of recycling this potent industrial byproduct. The brainchild of water fluoridation was Gerald Cox, a researcher with the Mellon Institute in Pittsburgh. He received a request to look at fluoride’s effects on teeth from Alcoa lab director Francis Frary, who was concerned about mounting lawsuits related to the fluoride pollution his plant produced.
Another motivation was the reality that disposing of fluoride waste from its aluminum plants was becoming increasingly costly for Alcoa. Previously, the Mellon Institute had been the leading defender of the asbestos industry, producing research showing asbestos was harmless and worker health problems were purportedly due to other causes. Using “science” as a smokescreen, the Mellon Institute was able to save the asbestos industry from financial catastrophe.
As a result of their success in using science to prop up the asbestos industry at that time, it makes sense Alcoa chose Cox and the Mellon Institute to craft a story around the perceived health benefits of fluoride. To ensure their success, Alcoa executives realized public opinion about fluoride had to be carefully and continuously manipulated.
In a bold move, they hired public-relations pioneer Edward Bernays, who later became known as the “father of spin,” to head the U.S. water fluoridation campaign. Using psychological principles targeted at what he called, the “mass mind,” Bernays was quite successful in attracting public support for the widespread consumer use of fluoride.2
Read the rest at the above URL
https://truthaboutfluoride.com/what-is-fluoride/
https://www.ahealedplanet.net/fluoride.htm
Introduction
This essay's first drafts, written in 1998, began with a several page chemistry narrative that presented some chemistry concepts. The original chemistry prelude was designed to help the reader understand the nature of fluoride, why it is used in industry how it is, why it is a waste product of certain industrial processes, and lays the groundwork for understanding why it was so important in the nuclear industry. Early readers felt that it was too much technical information for the lay audience, particularly to begin their reading experience. That prelude is now an addendum to this essay. Reading the prelude is not imperative for understanding this essay, but might make some issues clearer.
In short, fluorine is the most reactive element known to science ("reactive" means its affinity to bonding with other elements).[1] In nature, fluorine is found bonded to other elements, never found by itself, and is rarely found in its ionic state. The industrial processes of the 19th century created the most toxic pollutants that humanity had seen to that time...
Read the rest at the above URL
Let me just add that sugar is a sedative




![THE PAST 80 YEARS OF GLOBAL DECEPTION ☭ [BANNED DOCUMENTARY FROM THE 40ᵴ]](https://s3.us-central-1.wasabisys.com/altcast1/upload/photos/2025/03/ZDzBfe6Yg6APsb5aPbkz_28_9d9f8125a9c7031b288acc139bdc0086_image.png)
![THE NEW WORLD ORDER PLAN ☭ REVEALED IN FULL❗[THE MULTI PRONGED ATTACK EXPLAINED BY AI]](https://s3.us-central-1.wasabisys.com/altcast1/upload/photos/2025/03/WxvJIO4GxxnZnfahr7nN_13_0869f58ec895396aae3ab58ae68c0d1e_image.gif)
![THE BOOK OF ENOCH 📖 FULL AUDIOBOOK WITH TEXT [COMPLETE VERSION]](https://s3.us-central-1.wasabisys.com/altcast1/upload/photos/2025/03/I2dZMnr6dK4By4cUojBL_07_7a4427bff53fb76982af03a82c80a6ec_image.jpg)
![TOOTHPASTE IS MAKING YOU DUMBER ☠ [FLUORIDE IS A CARCINOGEN AND A NEUROTOXIN]](https://s3.us-central-1.wasabisys.com/altcast1/upload/photos/2025/03/xrr6PVU1nvDsEuq4kePj_03_216c9b2eb6f6632a8aea9c03e300fc59_image.png)





![THEY LIVE [LIKE C.H.U.D.] WAS A DOCUMENTARY❗](https://s3.us-central-1.wasabisys.com/altcast1/upload/photos/2025/01/rI5cdaYKCQ8Tlop5kHJg_25_9dc28fa56c5ddb5bf071406e294072ce_image.gif)

![BBC EXPOSED 🇬🇧 MARK COLLETT [FULL INTERVIEW 📼 COVERT RECORDING]](https://s3.us-central-1.wasabisys.com/altcast1/upload/photos/2025/01/kCW2MNcI3d3jtJa1iETB_18_bd14326326e151817691e542531aef84_image.jpg)



![WHITE TERROR DOCUMENTARY 卐 [A TERRORGRAM PRODUCTION]](https://s3.us-central-1.wasabisys.com/altcast1/upload/photos/2025/04/bpqPbIBZZiTcoX5S5THB_29_69e3217d8977cecf838fcb50c734b87c_image.png)
